CENDHY – COVID-19 vaccine: it's not a speed race

The World Health Organization reported in the first days of May that more than 100 candidate vaccines against the new coronavirus responsible for the most severe pandemic of recent decades, of which 7 or 8 are the most promising as they are the ones that are likely to deliver the best results in terms of their ability to generate immunity preventively.
But "creating" a vaccine isn't as easy or as fast as saying it, and there are reasons for this to be the case. Historically, vaccine development has taken many years, for example, for typhoid fever, a disease that reached relevance in the 19th century, it was until 105 years after the identifying agent that managed to develop the vaccine capable of combating the disease was identified. For measles, the process took more than 40 years and for the Hiv Human immunodeficiency viruses go 37 years without reaching a successful outcome.
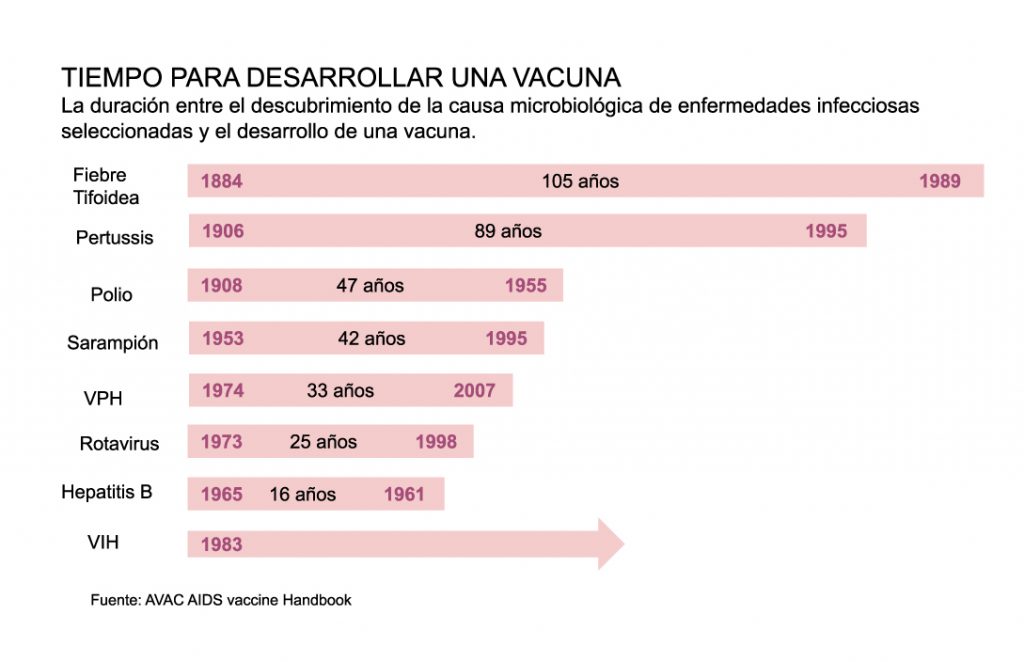
The mumps vaccine, –considered for now – to be the fastest in history, took 4 years from collection of biological samples to approval and licensing in 1967. This disease is caused by a paramyxovirus virus of which only one serotype is known, fact that facilitated the speed of the process. However, all indications are that the COVID-19 vaccine will arrive to take the throne from the vaccine for Mumps within the next few months.
 The Chilean experience
The Chilean experience
In our country, the respiratory syncytial virus vaccine in which Dr. Alexis Kalergis's research team works, Director of the Millennium Institute of Immunology and Immunotherapy (IMII) Pontifical Catholic University, began this effort the year 2004 and the culmination of this process is expected soon.
However, Dr.. Kalergis is one of many laboratories that has redirected its work to the development of a vaccine for COVID-19. The vaccine in development for this group consists of using antigens or fragments of coronavirus antigens, a strategy they chose for their ability to induce a favorable immune response for the elimination of the virus in the absence of excessive inflammation.
The use of SARS-CoV-2 antigens, molecules - usually proteins on the outside of viruses- who are specifically recognized by the host's immune system and trigger the defense, is a strategy equivalent to that already implemented by this research group in the successful development of the respiratory syncytial virus vaccine.
According to Dr.. Kalergis, the Chilean vaccine may be available for large-scale production within a minimum of 2 years, only if the conditions are met in the best possible scenario. Recently received a financial contribution from the Copec-UC Foundation to continue trials of the vaccine, which certainly means facilitating the process. However, the researcher is sensible in emphasizing that "these processes take time and require a number of preclinical and clinical studies that must comply with fairly strict regulations”, referring to the start of human tests estimated for 2021.
Normal times against the urgency of the pandemic
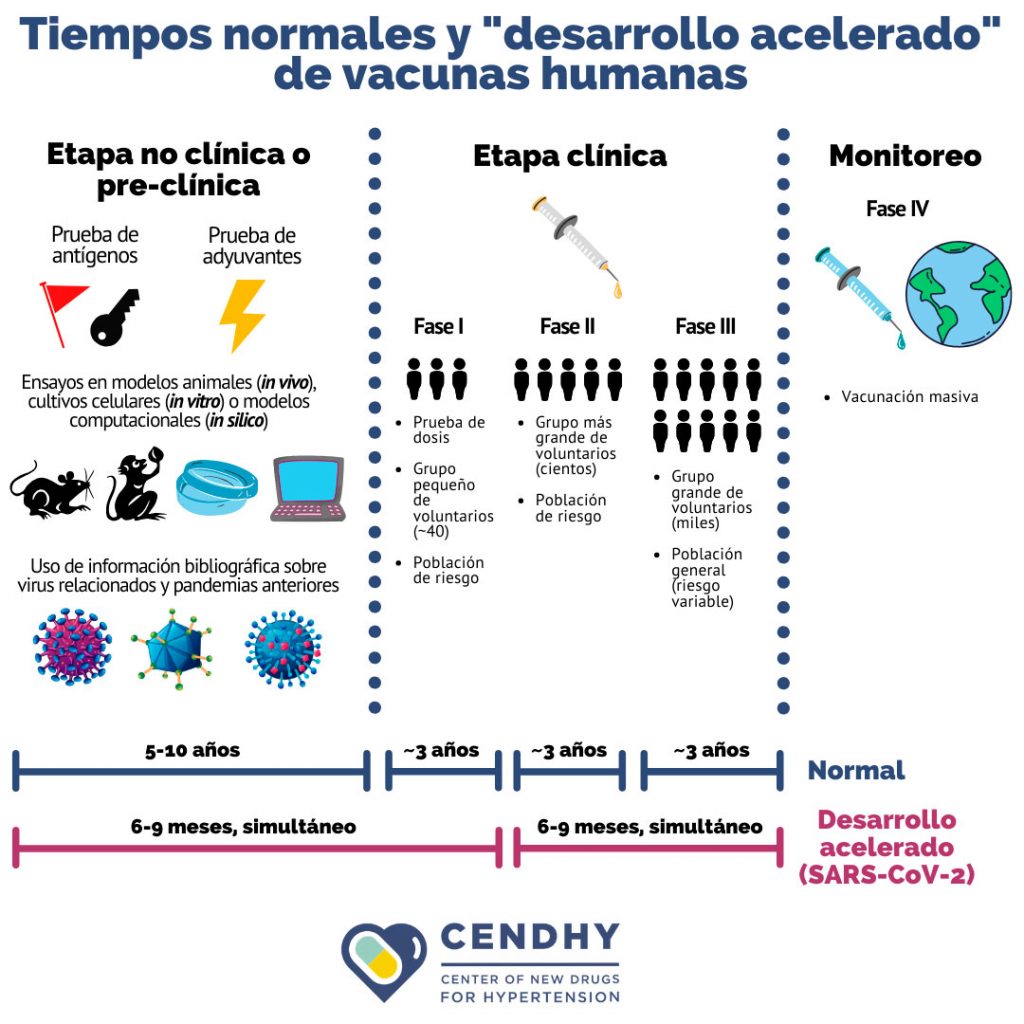
Vaccine development continues to be a long process, complex and linear that takes an average of 10 to 15 years, but in this case for COVID-19 the normal deadlines have been changed and what has been called accelerated development has been adapted, where research and regulatory process times are shortened, in addition to accelerating and even overlapping the experimental stages of clinical and preclinical trials. This adaptation does not mean that the safety or quality of the final product is reduced.
Along with efforts to best contain the spread of the virus, such as sanitary and confinement measures, hundreds, but thousands of scientific research teams around the world, are focused on finding a vaccine to tackle the pandemic and immunize the population. Although the hope of the public lies in a vaccine for COVID-19, researchers are clear that the development of a vaccine is not a speed race, but a true human pyramid that requires the cooperation and commitment of numerous actors, and that it is equally important to be able to ensure both the efficacy and safety of a long-awaited prevention element such as this vaccine.
The urgency for an early solution
At the beginning of June 6 million confirmed positive cases for this coronavirus were recorded worldwide, and more than 371 have been reported.000 deaths, reflecting the severity of the new pandemic we face since first case reported in China's Wuhan region in December 2019.
The disease caused by the new coronavirus, Called SARS-CoV-2, COVID-19 and produces flu-like symptoms: Fever, dry cough, shortness of breath, muscle pain and fatigue. In severe pictures it can lead to pneumonia, acute respiratory distress syndrome, even sepsis , exacerbated response to infection and septic shock, which leads some sick to death.
Why a vaccine?
First you need to answer another question: how a vaccine works? And to clarify this question, before we need to address certain points related to the human immune system, our agency's defense system, which is made up of specialized cells and diverse organs in our body.
The goal of vaccination is clear: Prevention. A vaccine simulates an infection but does not trigger the disease, what it does generate in the body is the onset of the immune response, that's why it refers to vaccination as part of the state programs population immunization. The human immune system acts in coordination through innate and adaptive responses, and vaccination points to the adaptive immune system, who "learns" to recognize invasive pathogens that are unknown to him, such as SARS-CoV-2, to prepare the organism for a new event of infection with the same pathogen.
It is important to understand that a vaccine does not give to the individual who receives it from shields to protect against the disease, nor is it a remedy that alleviates the disease. The way a vaccine delivers protection is that it promotes the body to generate – with its own raw material – its specific defenses for that infectious agent.
The path of coronavirus in the human body
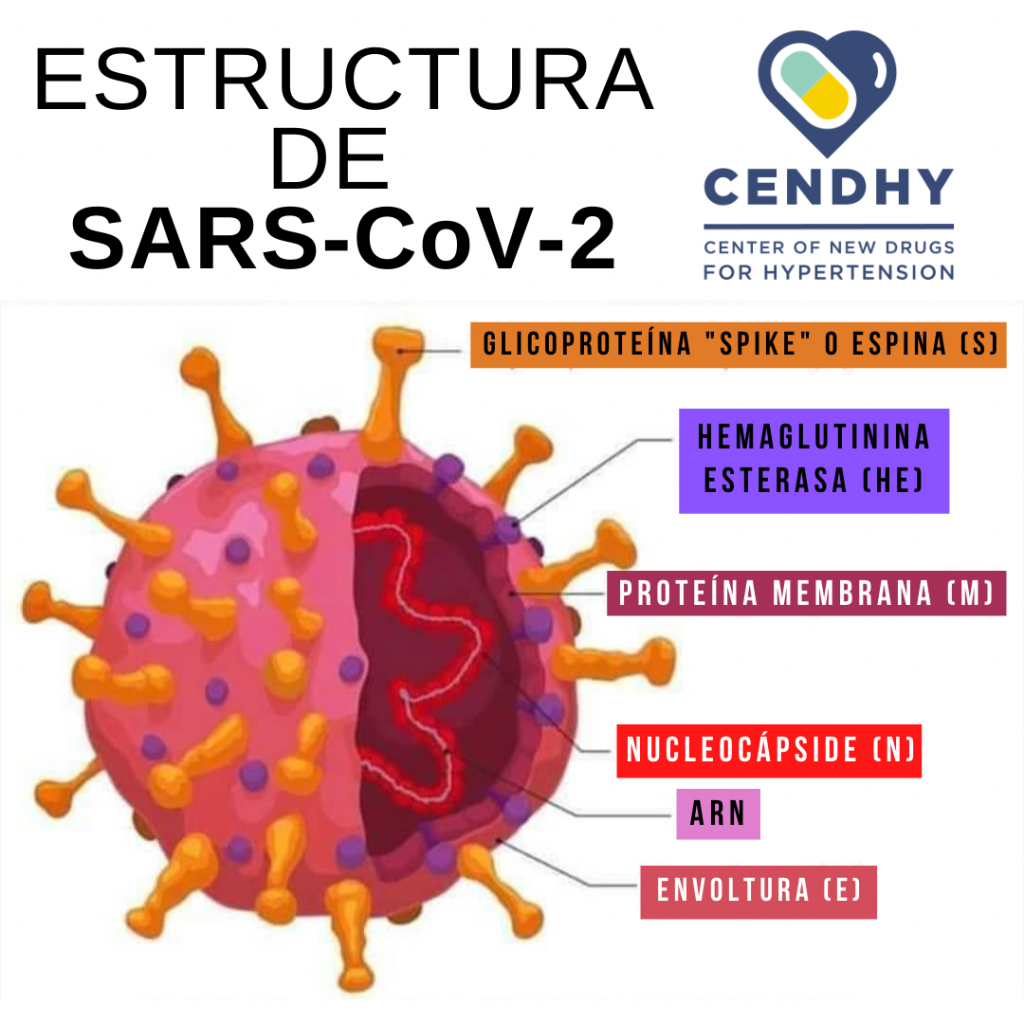
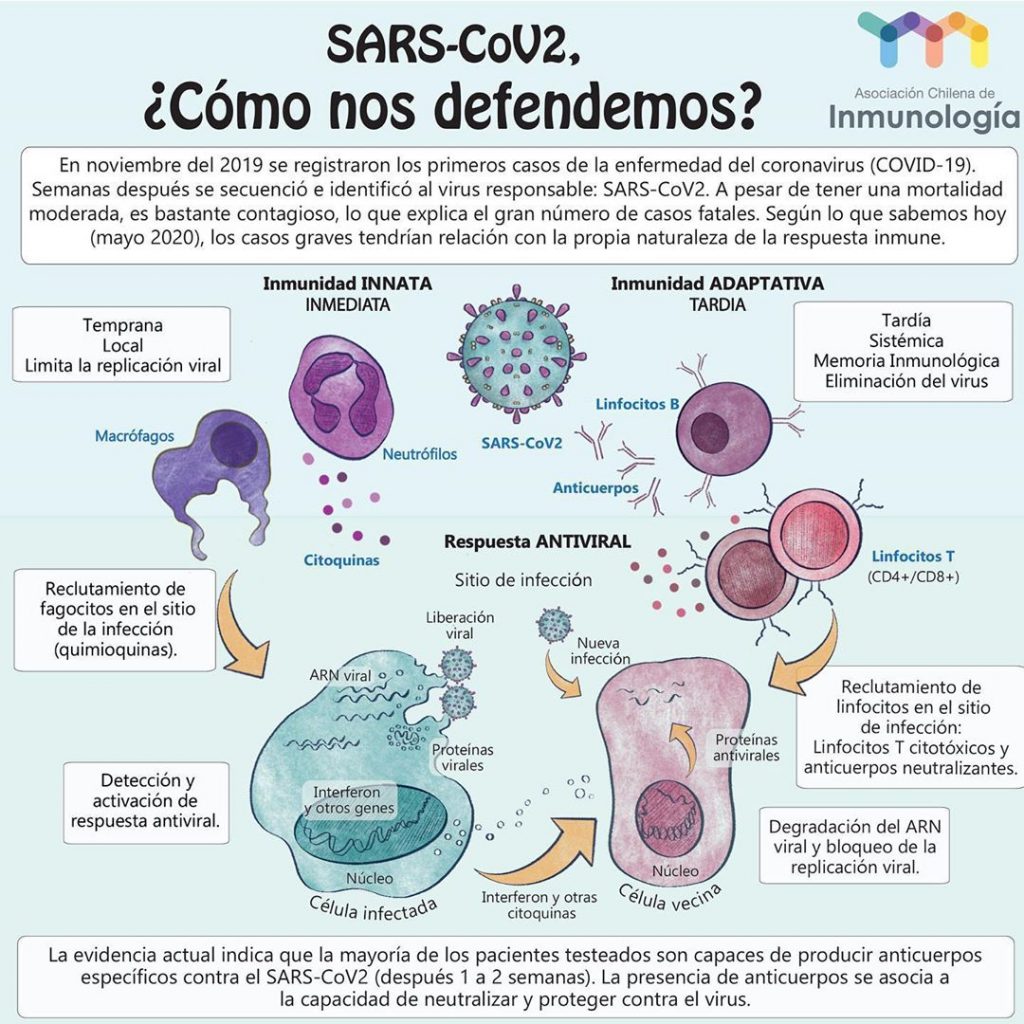
Faced with exposure to the new Coronavirus, our body fails to identify it and "lets it pass" freely through various ways of entry: Nose, Mouth, skin and any epithelium with mucous membranes that orients to the outside. SARS-CoV-2 is a virus whose genetic material is an individual strand of ribonucleic acid (Rna) What, unlike in human cells, it is not protected inside a closed compartment as is the cell nucleus. Another fundamental difference is that human genetic material is a double strand of deoxyribonucleic acid (DNA). Despite this apparent incompatibility, this coronavirus is able to break into our cells, in the nuclei of our cells, in our genetic material (DNA) and cause great damage.
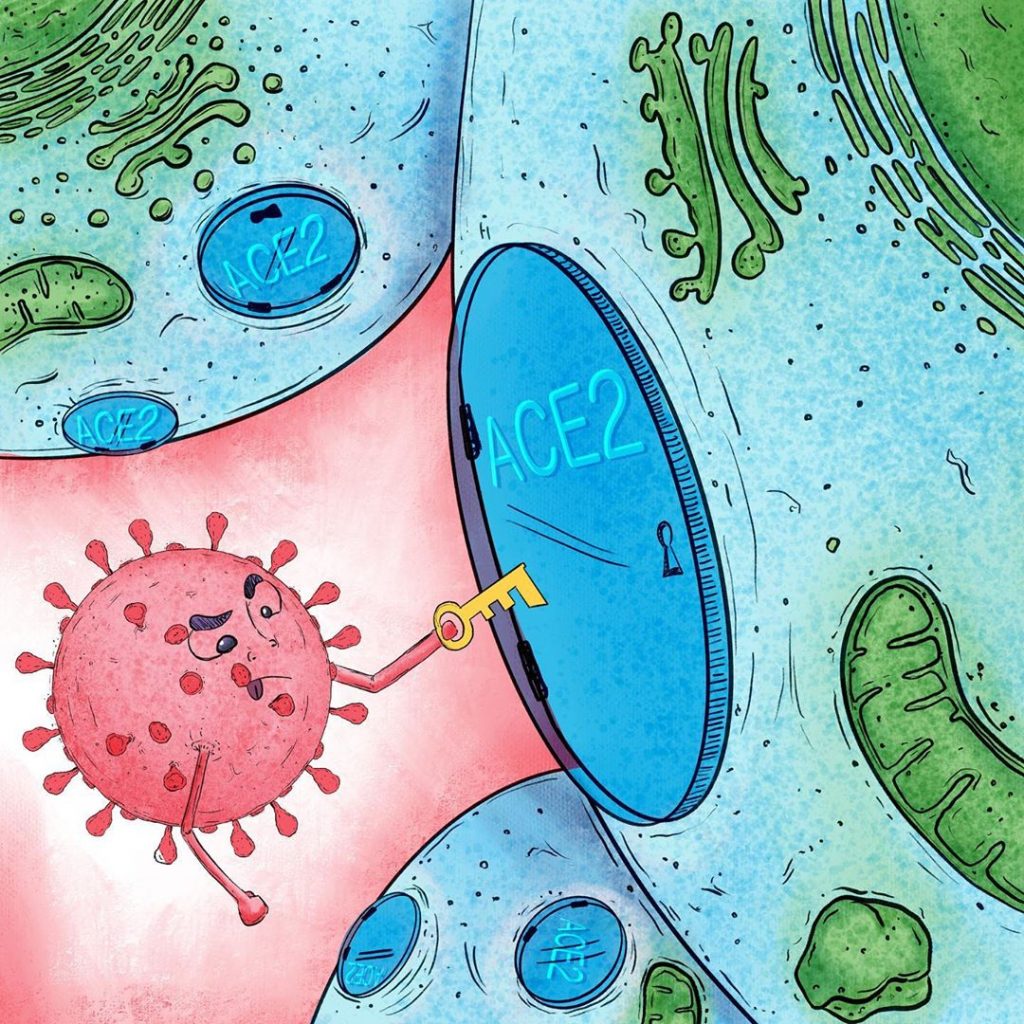
While the RNA of this virus is not found within a nucleus, coronavirus has an outer boundary known as wrapping, composed mainly of lipids and proteins. Particularly important are the famous spike protein or "thorn", and the M protein, both outward-facing. These two proteins are the main responsible for the entry of SARS-CoV-2 into circulation, as the virus binds to the Angiotensin II Converting Enzyme (ACE2) on the surface of human cells in numerous organs of the body, like lungs, kidneys and heart, and enters without much problem.
This process of recognition is often described, binding and anchoring ligands with their receivers using the "key and lock model" (“Lock and key”), where the key is one of the virus proteins, in this case the spinal protein, and the lock is the surface protein, ace2 receiver, allowing access to cells.
Once inside a human cell, coronavirus takes control of all available tools and begins to produce more viral particles, clones of himself, and to infect both nearby and distant cells. Immune response is initiated when a specialized type of cells, Antigen Presenting Cells (APC por sus siglas en inglés "Antigen Presenting Cells"), envelop the virus and expose on its own surface some important fragments of it to "get the attention" of other immune system cells and thus activate them, that's why they're said to "present" them.
These cells that are "called" APC are the "collaborating T cells", which in turn allow other branches of the immune response to be initiated: B cells or B lymphocytes produce antibodies that prevent the virus from infecting more human cells, as well as "marking" the virus to be recognized as a target of destruction. Thanks to this brand, "cytotoxic T cells" identify virus-infected cells and eliminate them, hence his name: "toxic to cells". That's right, in a simplified way, is how the immune response to a new or unknown infectious agent is carried out.
Vaccination and immune memory
Vaccination mimics an infection as it uses these important elements of the virus, we mentioned earlier, that "catches the eye" of immune system cells, trigger them and trigger the start of the immune response. The fundamental difference is that this imitation is in a controlled way and does not generate the disease, However, once the imitation of the infection "disappears", the body is left with a reservoir of memory lymphocytes that will "remember" how to fight the disease in the event that in the future it encounters the same infectious agent, what is known as immune memory. This memory is specific and can last several years, reactivates every time the body is again confronted with the same pathogen.
Vaccine development: "not so easy, not so fast"
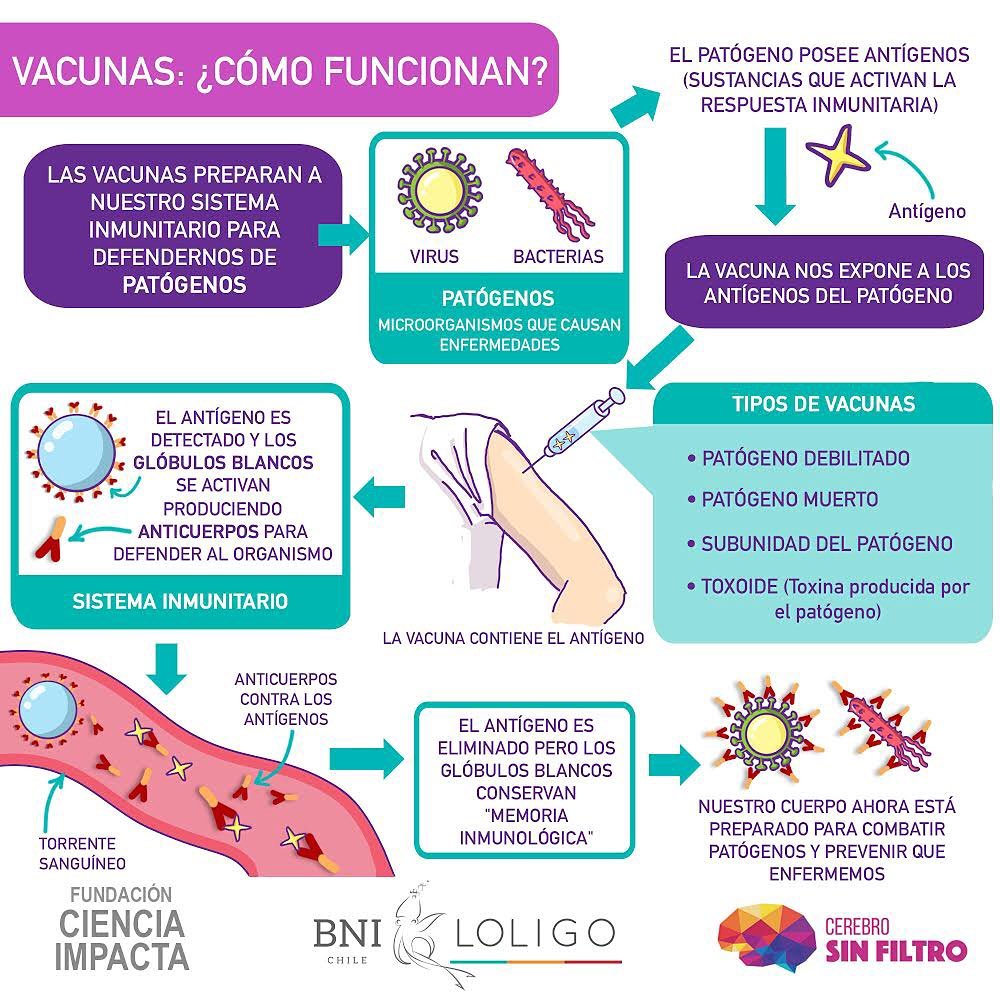
To develop a vaccine, you must first identify the microbiological agent that causes the disease, Study, characterize it, and then investigate the antigens that will be useful for a possible vaccine. An antigen is one of these "important elements that catch the eye of immune system cells, trigger them and trigger the start of the immune response" to which we have referred. The technical definition of antigen is "any substance that triggers the immune response".
All vaccines are intended to expose to our body an antigen that will not cause the disease, but will cause an immune response that will block or eliminate the pathogen if the person becomes infected next time. The presentation of the antigen is the milestone that triggers the immune response.
Post-R and development in the process of creating a new vaccine, details such as their regulations should be reviewed, then manufacture on a small scale with the corresponding quality control. The next step is preclinical testing on animal models (in vivo) to check that an immune reaction actually occurs and that the vaccine is not harmful, to finally begin clinical trials consisting of 4 consecutive stages: I, Ⅱ, III and IV, testing vaccines in humans.
Each stage must be properly exceeded and certified to advance to the next, that's why the total process of developing a vaccine is extensive in time, complex by the multiple instances of control, and linear where the stages are consecutive and follow a protocol order established for any manufacturer.
"The fastest vaccine ever"
Of the nearly 100 proposals for vaccines under development against SARS-CoV-2 by research teams in private companies, research centres, universities and laboratories around the world, researchers have studied different techniques, some of which have not been used in approved vaccines in the past.
At least nine groups have already started human safety trials according to WHO records, testing injectable vaccine formulations, while others are in the tests on animal models. Importantly, "on-the-run" companies have accelerated the process of developing vaccines in different ways: testing on several animal models at once, and in parallel with tests in small groups of people. Usually the process is to start in animal models and with those results progresses to human trials, to ensure that measurable immune response is obtained and that side effects are minimal. For this reason, researchers acknowledge that lack of time leads to higher risk.
However, studies have shown that SARS-CoV-2 does not mutate as fast as other viruses such as influenza or HIV, you have the confidence that a vaccine, once developed, can be effective and deliver protection for quite some time. Since the development of a vaccine for COVID-19 has taken the path of "accelerated development", Dan Barouch, Director of Vaccine Virology and Research at Beth Israel Deaconess Medical Center in Boston, Professor at Harvard University School of Medicine, and leader of one of the laboratories that is working on a vaccine for COVID-19, says that according to industry predictions by early 2021 an emergency vaccine or compassionate use could be available.
Barouch remembers that the vaccine for Ebola, which also sparked an emergency, it took 5 years to reach the population. A possible vaccine for COVID-19 is expected within one year, so the researcher concludes that "will be the development of a faster vaccine in history".
Among the most well-known and rated cases as promising are the Modern Therapeutics companies in Cambridge, Massachusetts, and Inovio Pharmaceuticals in Pennsylvania, both in the United States, preclinical and clinical trials in parallel.
Moderna announced on 18 May that phase 1 human trials yielded promising results, However, it's important to critically analyze this kind of news. The company reported that the test consisted of inoculating only 8 healthy volunteers and measuring their immune response through the production of antibodies, what immunology experts describe as Insufficient to ensure effectiveness. With these results, and in conjunction with the U.S.. National Institute of Allergy and Infectious Diseases, Modern advanced to phase 2 of its rehearsals.
The vaccine in development in the UK led by the University of Oxford and the company AstraZeneca, in charge of licensing, received about $1.2 trillion from the government American and is being compromised by September of this year. Just like that, Novavax is conducting clinical trials for the COVID-19 vaccine in Australia, and received a noun contribution of $388 million from a nonprofit initiative founded by Bill Gates. This vaccine, to be approved, would have a bit of Chile as its developers are currently researching the use of saponin, a substance of a similar structure to the soap produced by the Quillay tree, native to our country, as an indispensable component of the vaccine enhancer, called "Matrix-M".
It's not a speed race.
While a quick solution is needed, urgency cannot compromise people's safety and health. The analogies of "the race for the coronavirus vaccine" exclude an inherent aspect of scientific work, which is the verification of a hypothesis based on the concrete results, who arise from experimenting in a thorough way, rational and justified.
Accelerated development protocol ensures it doesn't compromise efficiency, safety, nor the quality of the final product that is a vaccine. Adapting normal development times exposes another aspect of scientific research: it's not an individual exercise, but it requires communication, cooperation and coordination between people, institutions and countries. Only in this way has it been possible to reformulate the development of a vaccine for COVID-19 and offer a solution in record time.
Developing a vaccine is not a speed race, it's more of a team sport or a human pyramid, where each level and each block are essential to build the entire pyramid, from the base to the top.
Not all vaccines are the same
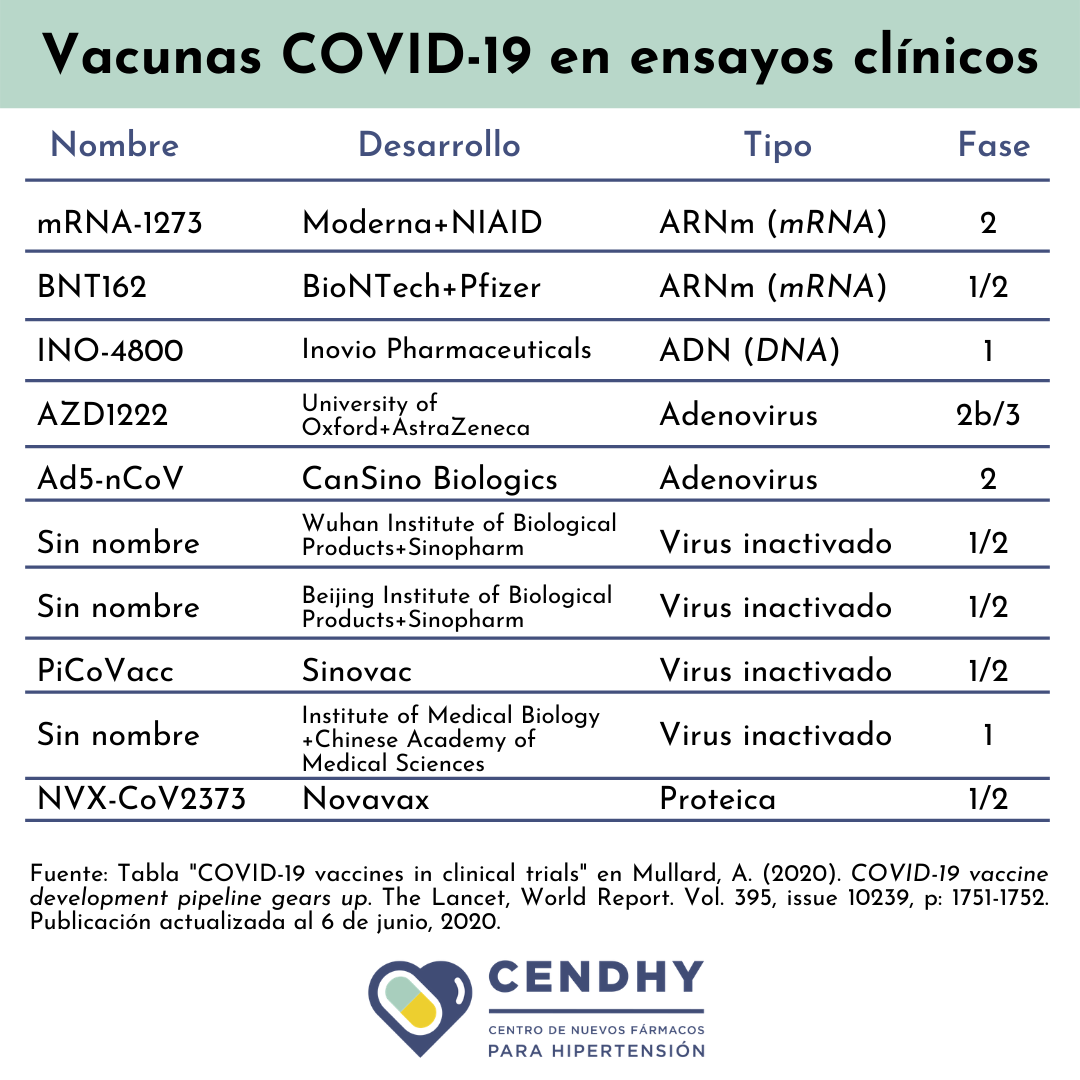
Many coordinated and distributed human teams worldwide are currently testing at least 8 types of vaccines for the new SARS-CoV-2 coronavirus. Within the types of vaccines in which work is, these use different approaches to fulfill the same purpose: in some cases they use the virus in full and in others they use parts of the virus to activate the immune response.
According to the press and in the words of the researchers themselves, the vaccines that "take the lead" in this metaphor of the speed race are basically the ones that are ahead and have presented preliminary results in their human trials. These include the Moderna Therapeutics candidate who is viral RNA; the joint proposal between the pharmaceutical company Pfizer and the company BioNTech which is also based on RNA; while the strategy chosen by the company Inovio Pharmaceuticals is DNA-based, just like the Harvard University working in partnership with Janssen Pharmaceutical of the multinational Johnson & Johnson. The last contender originating from the United States is the one proposed by the biotech company Novavax and funded in part by Bill Gates.
The advance English promise of the University of Oxford and AstraZeneca would be recombinant, as is the Ebola vaccine. Of this same nature is the vaccine under development in the company CanSino Biologics, in China. Its direct local competence is 3 viral type vaccines that employ inactivated SARS-CoV-2, and are under development at the Wuhan Institute of Biological Products, The Beijing Institute of Biological Products and the pharmaceutical company Sinovac Biotech. Since the latter, research leaders assured at the end of May that their CoronaVac vaccine has a 99% chances of being successful after passing the monkey trials and advancing to the second stage with more than 1000 human volunteers.
According to the latest update, 10 candidates are reported clinical stage, mostly performing phases 1 and 2 in parallel.
 Necessity over uncertainty
Necessity over uncertainty
To conclude, it is important to understand that vaccines can be developed using different techniques, but the end is the same: create a vaccine capable of generating immunity in the population to deal with this pandemic. Although this process can usually take more than 10 years, in this emergency case to bend COVID-19, stages are accelerating, both experimental and regulatory.
With accelerated processes like these it is normal for doubts and mistrust to arise: Is it safe?, when will the vaccine arrive?, Do I need to get vaccinated if I had the disease?, who gets immune?, can I get it again or spread? After a review of studies on common colds some researchers initially assumed that those recovered from SARS-CoV-2 infection may be protected from reinfection for a certain period of time, what calls them "immune". Regarding these assumptions, Michael Diamond, viral immunologist at the University of Washington in Missouri, United States, states that they should be "backed by evidence". Diamond also states that "we don't know much about the immunity of this particular virus" [14], against which you must be cautious and patient until you get certainties.
Another of the most common concerns is how long the immunity granted by the vaccine would last. According to Stanley Perlman, virologist specializing in coronavirus at the University of Iowa: "there is no good evidence to indicate lasting immunity". In response to this question, Diamond hopes more can be found on infection in animal and human studies, and that "perhaps this accelerated way of developing a vaccine is not the most efficient but may be the most convenient" [14].
The immediate need is to prevent more deaths and severe pictures of the disease, stop the rise of the contagion curve and protect the population, and in the face of that the consensus decision among experts from various scientific areas, medical specialists and public health authorities, is to act intelligently about uncertainties, opting for accelerated vaccine development in a context of interdisciplinary collaboration.
